The addition of nivolumab to first-line chemotherapy has led to survival benefit in patients who have metastatic gastroesophageal adenocarcinoma with a programmed death ligand 1 combined positive score of 5 or greater. 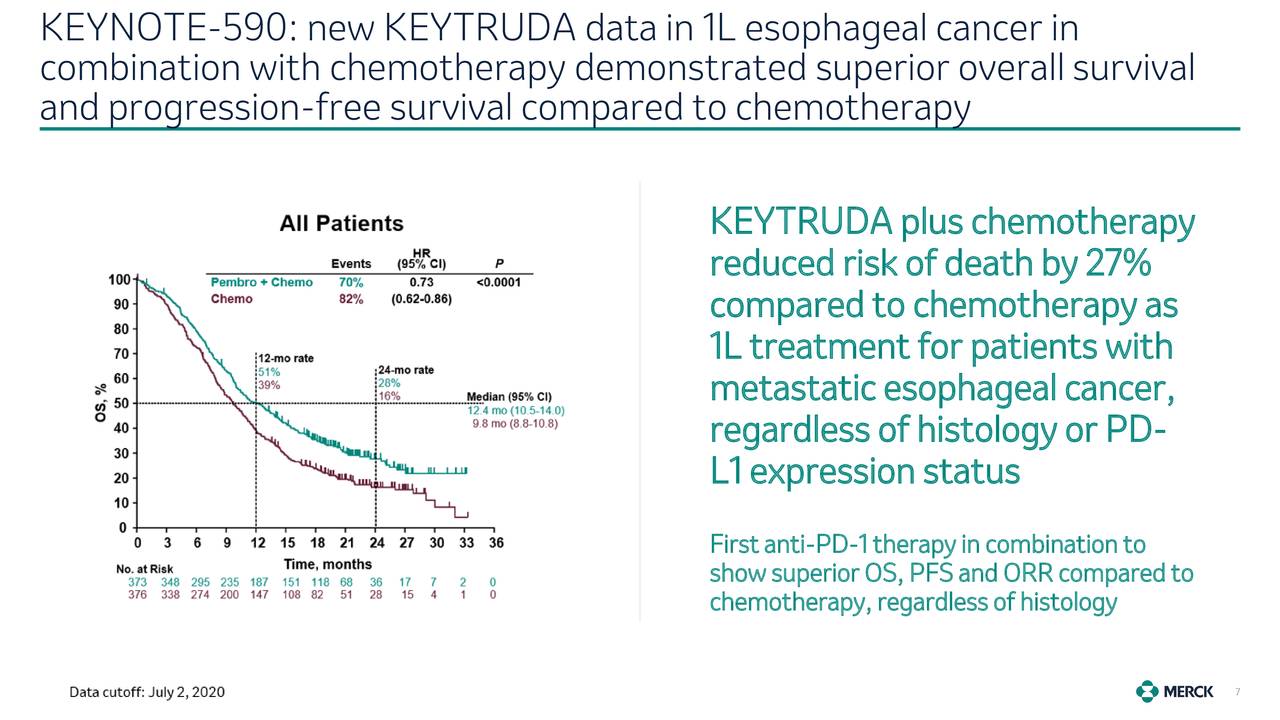
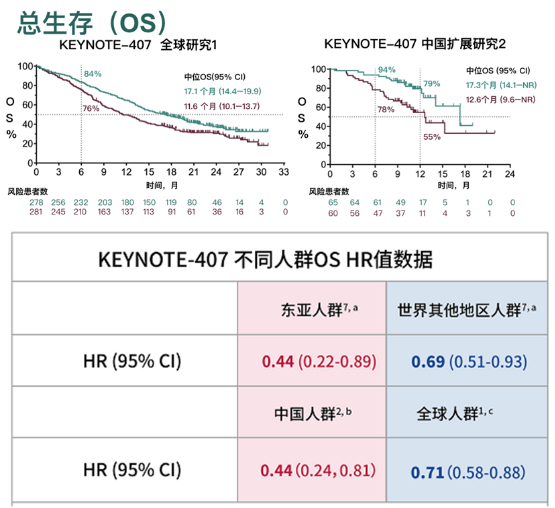
There was statistically significant improvement in outcome in the CPS 10 and greater population as well as all comers.Abstract: The treatment landscape for gastroesophageal adenocarcinomas has significantly changed over the last year. This study demonstrated an improvement in overall survival from 8 to 9 months to about 13 months. As such, it has been incorporated into care for this population. Based on this study, the FDA approved the addition of pembrolizumab to chemotherapy for patients with adenocarcinoma and squamous cell carcinoma in combination with 5-FU–platinum–based treatment. Furthermore, this was demonstrated significantly in patients who had esophageal squamous cell carcinoma. This was demonstrated in all comers and patients with a PD-L1 CPS of 10 or greater. This trial demonstrated that patients who received chemotherapy, 5-FU and platinum-based treatment plus pembrolizumab, had a superior outcome in terms of overall survival compared with chemotherapy alone. About 50% of the patients were accrued from Asia. That was approximately 70% of the population. The majority of patients who participated in the trial had squamous cell carcinoma. The coprimary end points of the study were overall survival and progression-free survival in all comers and patients who had a PD-L1 CPS of 10 or greater. Patients were randomized to chemotherapy vs chemotherapy plus pembrolizumab. 
Syma Iqbal, MD: KEYNOTE-590 was a randomized phase 3 trial for patients with untreated esophageal cancer, both adenocarcinoma and squamous cell carcinoma. 
Now Viewing EP: 3.KEYNOTE-590 Trial Impacting Treatment Landscape for ESCC and Upper GI Cancer EP: 4.Overview of CheckMate Trials 648 and 649 EP: 5.Aligning ESCC and GI Cancer Treatment Strategies With Current Guidelines EP: 6.Quality of Life Impacted by Treatment of ESCC and Upper GI Cancer EP: 7.Optimizing Treatment Pathways for Patients with ESCC and GI Cancer EP: 8.The Evolving Treatment Landscape of Upper GI and Esophageal Cancer EP: 9.Key Cost Drivers in Treating Gastroesophageal Cancers EP: 10.Unmet Needs for Patients With Esophageal and Upper-GI Cancers EP: 11.Role of Immune Checkpoint Inhibitors in Gastroesophageal Cancer Treatment Landscape EP: 12.Decision-Making Within a Rapidly Evolving Field Where Labeling May Differ From Current Clinical Guidelines EP: 13.Determining Individual Treatment Sequencing for Patients with Gastroesophageal Cancer EP: 14.Selecting Optimal Gastroesophageal Treatment: Chemotherapy, Immunotherapy, and Combined Therapies EP: 15.New Gastroesophageal Treatment Therapies Impacting Patient Quality of Life EP: 16.Defining Appropriate Response to Gastroesophageal Cancer Treatment EP: 17.Immunotherapy Considerations for Upper GI and Esophageal Cancer Treatment Pathways EP: 18.Evaluating Gastroesophageal Cancer Pathway Decisions EP: 19.Cost-Effective Analyses for Upper GI and Esophageal Cancer Treatment EP: 20.Patient-Support Services and Programs for Patients With Gastroesophageal Cancer
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
